

学习资源
科普文章
免疫哲学(二):免疫与肿瘤的微妙关系[1]
免疫学是当今生物和医学科学中最核心和最具活力的领域之一,翻阅最新顶尖科学或医学期刊时,甚至阅读关于新冠和癌症等科普微信文章时,我们会产生免疫学无所不在的印象。为什么免疫学在我们的科学和日常生活中变得如此重要——为什么会启迪我们哲学的思考?为此我们为读者推荐一篇名为免疫哲学(Philosophy of Immunology)文章[1],作者为法国国家科学研究中心和波尔多大学(CNRS & UniversityOf Bordeaux)教授Thomas Pradeu。他关于免疫系统与微生物、肿瘤、神经系统等微妙关系的描述非常精彩,并富于哲学思考,强力推荐感兴趣的读者可以下载原文阅读(下载链接为:https://www.cambridge.org/core/elements/philosophy-of-immunology/06F0C341035299674EECF0406E5D8E31)。我们会将这篇文章中关于免疫与肿瘤相互关系的内容分二期,第一期请点击,肿瘤与免疫相互关系的“3E”理论:消除“elimination”-平衡“equilibrium”-逃逸“escape”[1]
以下是第二期内容,因为英文原文写得很浅显易懂,所以将其保留,并附上中文翻译供大家参考:
Immune–Cancer Interactions: Current Views and Clinical Applications
免疫-癌症相互作用:当前观点和临床应用
Today, the evidence showing that the immune system plays a central role in cancer development (either preventing or promoting cancer) is overwhelming. Many cellular and molecular immune components are involved in this process of restriction or promotion of cancer. Additionally, these interventions of the immune system can occur at different levels in or around the tumor (including genes, cells, and the tumor microenvironment) and at every step of its progression, from initiation to neoplastic progression to metastasis. For example, mouse models showed that tumor-associated macrophages promote angiogenesis and tissue remodeling, thereby favoring tumor growth. Clinical studies show that extensive tumor-associated macrophage infiltration positively correlates with cancer metastasis and poor clinical prognosis.
今天,有大量证据表明免疫系统在癌症发展(预防或促进癌症)中起着核心作用。 许多细胞和分子免疫成分都参与了这个限制或促进癌症的过程。此外,免疫系统的这些干预可以发生在肿瘤内部或周围的不同水平(包括基因、细胞和肿瘤微环境),并且发生在其进展的每一步,从起始到肿瘤进展再到转移。例如,小鼠模型显示肿瘤相关巨噬细胞促进血管生成和组织重塑,从而有利于肿瘤生长。临床研究表明,广泛的肿瘤相关巨噬细胞浸润与癌症转移和不良临床预后呈正相关。
Investigations about the role of the immune system in cancer progression have led to remarkable clinical applications. For example, several groups have shown how, in a diversity of cancers, infiltration by certain immune components had a better prognostic value than more traditional approaches. But most crucially, since the 2010s, the knowledge accumulated over decades about immune-mediated control of cancerous tumors has turned into specific clinical applications called “cancer immunotherapies,” which many have described, rightly or wrongly, as “revolutionary”. After extremely encouraging results obtained in melanoma and a few other cancer types at the beginning of the 2010s, evidence in favor of the success of immunotherapies (increasingly, in fact, a combination of immunotherapies) in several cancers has accumulated. In a number of situations, the results have been unprecedented and sometimes even spectacular, especially in cases of previously incurable cancers, raising much enthusiasm. Immunotherapies are diverse but recently immune checkpoint inhibitors have been particularly explored.
关于免疫系统在癌症中的研究已经有了重要的临床应用。例如,几项研究都表明,在多种癌症中,某些免疫成分的浸润具有更好的预后价值。但最重要的是,自 2010 年代以来,几十年来积累的关于免疫控制癌症的知识已经转化为“癌症免疫疗法”的临床应用,许多人将其描述为“革命性的”。在 2010 年代初在黑色素瘤和其他一些癌症类型中取得了极其令人鼓舞的结果,其中免疫检查点抑制剂最为受到关注。
The most significant recognition of the work done in this area is undoubtedly the 2018 Nobel Prize in Physiology/Medicine awarded to James P. Allison and Tasuku Honjo “for their discovery of cancer therapy by inhibition of negative immune regulation,” which is centered on the blockade of immune checkpoints. This has definitely convinced researchers working in all areas of cancer investigation and the lay public that it is indispensable to pay attention to the role of the immune system in cancer. It comes as no surprise that many newspapers have discussed these medical advances, if perhaps sometimes hyperbolically.
对这一领域所做工作最重要的认可无疑是授予 James P. Allison 和 Tasuku Honjo 的 2018 年诺贝尔生理学/医学奖,“以表彰他们通过抑制负性免疫调节来治疗癌症的发现”,其重点是对于抑制性免疫检查点的阻断。这无疑使癌症科研领域和普通公众相信,关注免疫系统在癌症中的作用是必不可少的。毫不奇怪,许多媒体已经讨论了这些医学进步,即使有时可能是夸张的。
Are immunotherapies really revolutionary, and what do they tell us about immune–cancer interactions? From a strictly medical point of view, this enthusiasm is justified, although we should keep in mind that some important limits exist. The first limit is the low percentage of responders: less than 15 percent on average, though it depends on tumor type and on the category of immunotherapy. (Future research likely will significantly extend the proportion of responders.) A second limit is the existence of sometimes significant adverse effects (immunotherapies, in particular, increase the level of inflammation and autoimmune responses, which can lead to colitis, hepatitis, etc.). A third limit is the currently exorbitant cost of some treatments.
免疫疗法真的是革命性的吗?它们告诉我们关于免疫-癌症相互作用的什么信息?从严格的医学角度来看,对于肿瘤免疫疗法的积极肯定是有道理的,尽管我们应该记住,它的一些重要的局限性。第一个限制是治疗应答者的百分比很低:平均不到 15%,尽管这取决于肿瘤类型和免疫疗法的类别。(未来的研究方向是如何提供患者应答率)。第二个限制是有时存在显着的副作用(特别是免疫疗法,会增加炎症和自身免疫反应的水平,这可能导致结肠炎、肝炎等)。第三个限制是目前某些治疗的高昂费用。
From a conceptual viewpoint, therapies based on immune checkpoint inhibition constitute indeed a radical change in perspective. At least two important features of immune checkpoint therapies are worth emphasizing. First, the target of the treatment is the immune system, not the tumor itself (as was the intention with traditional treatments such as surgery, chemotherapy, and radiotherapy – although, in fact, some of them are now known to act at least in part via stimulation of the immune system). Second, the objective is to break the state of immune tolerance that has been established between the tumor and the immune system in the local tissue – partly as a consequence of the chronic expression of cancer antigens. More precisely, the aim with immune checkpoint blockers is to downregulate inhibitory signals in tumor–immune interactions. This constitutes a move from enhancement of the immune system to “normalization” of the immune system: in immune checkpoint inhibitor-based immunotherapies, especially with anti-PD-L1, the aim is not, strictly speaking, to boost the immune system beyond its normal rate of activation but to restore a local context in which the immune system will be able to act as it normally does.
从概念的角度来看,基于免疫检查点抑制的疗法确实构成了概念的根本变化。免疫检查点疗法至少有两个重要特征值得强调。首先,治疗的目标是免疫系统,而不是肿瘤本身(这往往是手术、化学疗法和放射疗法等传统疗法的对象——尽管事实上,现在已知其中一些疗法至少部分通过刺激免疫系统起作用)。其次,目标是打破局部组织中肿瘤和免疫系统之间建立的免疫耐受状态——部分原因是癌症抗原的慢性表达。更准确地说,免疫检查点阻断剂的目的是下调肿瘤免疫相互作用中的抑制信号。这构成了从增强免疫系统到免疫系统“正常化”的转变:在基于免疫检查点抑制剂的免疫疗法中,尤其是使用抗 PD-L1 时,严格来说,其目的不是增强免疫系统,使其超出其正常水平的正常激活状态,而是要恢复免疫系统在局部环境中的正常状态。
Together, basic studies about the role of the immune system in cancer and clinical studies in the domain of immunotherapies also have contributed to an important change in perspective about what cancer is and how it develops. It is increasingly recognized that tumor-centric views of cancer (with genetic mutations seen as the main cause of cancer) are insufficient: to understand (and cure) cancer, it is essential to consider not only the tumor itself but also the tumor environment. The tumor environment includes the tissue context located at the vicinity of the tumor (sometimes called the tumor microenvironment), but also elements located quite remotely from the tumor in the organism (such as some immune-associated organs and the microbiota, which recently has been proven to influence cancer progression and therapies). Even authors who initially focused on intrinsic molecular aspects of cancer development have later emphasized the importance of the tumor microenvironment. Targeting the tumor microenvironment also offers enriched therapeutic strategies. There is a growing consensus that the immune system plays a crucial role in the tumor microenvironment. In fact, given the centrality of immune components in the organization of, and control over, the local tissue, it seems reasonable to say that every cancer involves the immune system, which necessarily intervenes, at one point or another, in the shaping of the local context that enables the tumor to emerge, grow, and perhaps spread.
总之,关于免疫系统在癌症中的作用的基础研究和免疫疗法领域的临床研究也促成了关于癌症是什么以及它如何发展的观点的重要变化。人们越来越认识到,以肿瘤为中心的癌症观点(基因突变被视为癌症的主要原因)是不够的:要理解(和治愈)癌症,不仅要考虑肿瘤本身,还要考虑肿瘤环境。肿瘤环境包括位于肿瘤附近的组织环境(有时称为肿瘤微环境),但也包括生物体中距离肿瘤相当远的元素(例如一些免疫相关器官和微生物群,最近已被 证明影响癌症进展和治疗)。即使是最初关注癌变细胞内在分子机制的研究者后来也强调了肿瘤微环境的重要性。针对肿瘤微环境也提供了丰富的治疗策略。人们越来越一致认为,免疫系统在肿瘤微环境中起着至关重要的作用。事实上,鉴于免疫在局部组织的构成和调控的核心地位,似乎可以合理地说,每一种癌症都涉及免疫系统,免疫系统必然会在某一时刻会参与到促使肿瘤出现、生长并可能扩散的局部环境的形成。
Role of the Immune System in the Breakdown of Biological Individuality That Characterizes Cancer
免疫系统在以癌症为特征的生物个体崩溃中的作用
考虑到所有这些关于免疫系统的肿瘤限制和肿瘤促进作用的讨论,我们现在可以回到我们开始的问题。免疫系统在预防以癌症为特征的个体崩溃中到底起什么作用?相对应的,免疫系统如何参与生物个体的解体?
The idea that cancer constitutes a breakdown of biological individuality is widespread in the scientific and philosophical literature. Biologist Leo Buss was instrumental in showing that biological individuality in multicellular organisms must be understood as an outcome of evolution, by which, on several occasions in life’s history, some cells aggregated and cooperated, and in which emerged some control mechanisms over cells that would proliferate at the expense of the whole organism. Buss takes cancer as an example of a decohesion of the biological individual, in which cancer cells are “re-individualized” in a way that becomes harmful to the multicellular organism. This idea has subsequently been explored by several biologists and philosophers of biology, often inspired by the study of clonal evolution at the cell level in cancer. For example, Godfrey-Smith labels as “de-darwinization” the process by which a higher-level individual prevents proliferation of lower-level individuals. From that point of view, cancer cells appear as a result of a “re-darwinization” at the cell level.
癌症使生物个体崩溃的观点在科学和哲学文献中广为流传。生物学家 Leo Buss 在证明多细胞生物体的生物个体性必须被理解为进化的结果方面发挥了重要作用,在生命的历史中,多个细胞聚集和相互作用,形成多细胞生物个体,并在个体中出现了一些控制细胞增殖的机制 ,以避免细胞“外溢”,而“侵害”整个生物体。Buss 将癌症作为生物个体退化的一个例子,其中癌细胞以对多细胞生物个体有害的方式“重新个体化”。这个想法随后被几位生物学家和生物学哲学家探索,通常受到癌症细胞水平克隆进化研究的启发。例如,戈弗雷-史密斯将高级个体阻止低级个体增殖的过程称为“去达尔文化”。从这个角度来看,癌细胞的出现是细胞水平“达尔文进化论”的结果。
However, the mechanistic details by which the multicellular organism exerts control over cancer cells have remained vague. Michod cites programmed cell death and the immune system as the two main “policing mechanisms” in the multicellular organism, but he does not give any detailed explanation about how they work. An important lesson of what has been said in this section is that immunological surveillance constitutes a convincing example of a mechanistically precise process of maintenance of cohesiveness in the organism. Thus, it offers an important contribution to this long-standing debate concerning de-darwinization in cancer. The details of how immune-mediated control works are well documented: in the elimination phase of immunoediting, myriad immune cells and molecules (macrophages, dendritic cells, NK cells, γδ T cells, CD4 and CD8 T cells, IFN-γ, among many others) contribute to the destruction of the tumor. In addition, immune-mediated control contributes to coordinate other control instruments, such as apoptosis and angiogenesis.
然而,多细胞生物个体对癌细胞施加控制的机制细节仍然模糊。 Michod 将程序性细胞死亡和免疫系统列为多细胞生物个体中的两个主要“监管机制”,但他没有对它们的工作原理给出任何详细解释。本节中所说的一个重要教训是,免疫监视构成了一个令人信服的例子,说明了机体内聚性维持的机械精确过程。因此,它为这场关于癌症去达尔文化的长期争论做出了重要贡献。免疫介导控制如何工作的细节有据可查:在免疫编辑的消除阶段,无数免疫细胞和分子(巨噬细胞、树突状细胞、NK 细胞、γδ T 细胞、CD4 和 CD8 T 细胞、IFN-γ 及其他)有助于破坏肿瘤。此外,免疫介导的控制有助于协调其他控制工具,例如细胞凋亡和血管生成。
This, however, strengthens rather than solves the paradox: if immune-mediated restriction is one of the main mechanisms ensuring the cohesion of the organism, we need an explanation for why in some circumstances the immune system favors tumor development (through the escape phase of immunoediting and/or through repair mechanisms that contribute to creating a favorable tissue environment for the tumor). To better understand this phenomenon, I propose here an extended view of immune-mediated cohesion and decohesion.
The traditional view about immune-mediated cohesion, as represented in classically defined immunological surveillance, is that the immune system can eliminate abnormal cells such as cancer cells (Figure 4.2).
然而,这加强了而不是解决了这个悖论:如果免疫介导的限制是确保有机体凝聚力的主要机制之一,我们需要解释为什么在某些情况下免疫系统有利于肿瘤的发展(通过逃逸阶段 免疫编辑和/或通过有助于为肿瘤创造有利组织环境的修复机制)。为了更好地理解这种现象,我在这里提出了免疫介导的凝聚力和去凝聚力的扩展观点。
关于免疫介导的凝聚力的传统观点,如经典定义的免疫学监视(“immunological surveillance”)所代表,认为免疫系统可以消除癌细胞等异常细胞(图 4.2)。
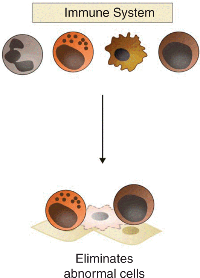
Figure 4.2 The traditional view of immune-mediated cohesion, as proposed by the immunological surveillance hypothesis. According to this view, the immune system directly eliminates abnormal cells, such as cancer cells (in red) in the tumor.
(Figure drawn by Wiebke Bretting).
图 4.2 免疫监视假说提出的免疫介导凝聚力的传统观点。根据这种观点,免疫系统直接清除异常细胞,例如肿瘤中的癌细胞(红色)。
(图由 Wiebke Bretting 绘制)。
This view, however, is too narrow, because it neglects the diversity of activities in which the immune system is involved, which include not only defense but also development, tissue repair, clearance of debris, and maintenance of tissue homeostasis, among others. In cancer, many of these immunological activities are found. This leads to a much richer view of immune-mediated cohesion and decohesion in cancer (Figure 4.3). In this view, the immune system plays a major role in regulation of the organization of the local tissue, and, together, the immune system and the tissue realize different activities, including the elimination of abnormal cells, but also the containment of abnormal cells (in that case, cells are not destroyed, they are simply kept under control, which limits the damage they can do and/or their capacity to spread), the maintenance of chronic elements present in the local environment (most of the time, these chronic elements are normal self components of the organism, but chronically present tumor constituents can also lead to active maintenance of the tumor by the immune system, which progressively sees these elements as normal) (Pradeu et al. 2013; Pauken and Wherry 2015), and the repair of the local tissue (in physiological conditions, this repair is a necessary process insuring the integrity of the tissue, but in the context of cancer repair mechanisms can favor cancer progression).
然而,这种观点过于狭隘,因为它忽略了免疫系统生物学功能的多样性,其中不仅包括防御,还包括发育、组织修复、碎片清除和组织稳态维持等。在癌症中,发现了许多这些免疫活性。这导致了对癌症中免疫介导的凝聚和去凝聚的更丰富的认识(图 4.3)。在这种观点下,免疫系统在调节局部组织的组织中起主要作用,并且免疫系统和组织一起实现不同的生物学功能,包括消除异常细胞,也包括异常细胞的遏制(在那种情况下,细胞不会被破坏,它们只是受到控制,这限制了它们可以造成的损害和/或它们的传播能力),以及对于微环境中某些慢性元素(chronic elements)的“容忍”和耐受(大多数时候,这些元素是生物体的正常自身成分,但肿瘤的长期存在也会导致免疫系统逐渐将肿瘤视为“正常”元素,并主动维持)(Pradeu 等人 2013 年;Pauken 和 Wherry 2015 年),以及局部组织的修复(在生理条件下,这种修复是确保组织完整性的必要过程,但在癌症修复机制的背景下可能有利于癌症进展)。
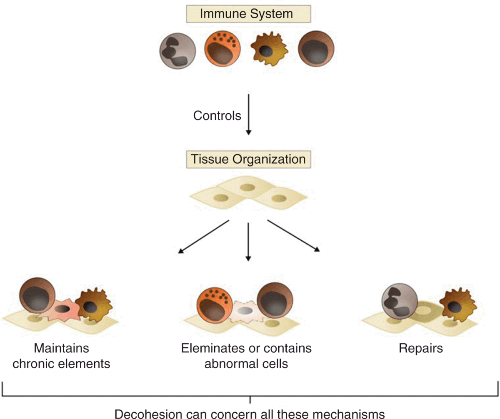
Figure 4.3 A richer view of immune-mediated cohesion and immune-mediated decohesion in cancer. In this view, the immune system controls tissue organization and, together, the immune system and the local tissue can exert a variety of cohesion-promoting activities, including the elimination of abnormal cells, but also the containment of abnormal cells, the maintenance of chronic elements, and tissue repair. All these activities (not just elimination), in pathological conditions, can promote decohesion of the organism.
(Figure drawn by Wiebke Bretting).
图 4.3 癌症中免疫介导的凝聚力和免疫介导的去凝聚力的更丰富视图。从这个观点来看,免疫系统控制着组织构成,并且免疫系统和局部组织一起可以发挥多种凝聚力促进功能,包括消除异常细胞,也包括异常细胞的遏制,维持慢性元素和组织修复。在病理条件下,所有这些活动(不仅仅是消除)都可以促进生物个体的分解。
(图由 Wiebke Bretting 绘制)。
The crucial point here is that decohesion as seen in cancer can concern all these different activities, not just elimination: the immune system, which in most cases prevents cancer progression by elimination, containment, maintenance, and repair, can in some circumstances promote cancer progression because of deregulated elimination, containment, maintenance, and/or repair. Moreover, all these activities must be understood diachronically: they do not all intervene at the same time, and the immune system can switch from one effect to the other (for example, it can initiate the destruction of abnormal components and later contribute to their maintenance because they have become chronically expressed in the tissue).
这里的关键点是,在癌症中看到的去凝聚力可以涉及所有这些不同的生物学功能,而不仅仅是消除:免疫系统在大多数情况下通过消除、遏制、维持和修复来防止癌症进展,但在某些情况下,又通过解除管制的消除、遏制、维护和/或修理,从而促进癌症进展。此外,必须从历时角度理解所有这些生物学过程:促癌或抑癌作用不会同时发生,免疫系统可以从一种作用切换到另一种作用(例如,它可以启动对于异常成分的破坏,然后又因为它们已经在组织中长期表达,又主动维系它们)。
Does the immune system dysfunction when it promotes cancer progression via a deficit in elimination or containment and/or via maintenance processes and/or via repair processes? I suggest distinguishing two situations here. The first situation corresponds to a dysfunctional immune system. An organism, either structurally or provisionally (e.g., after a treatment with immunosuppressive drugs), can have a defective immune system (e.g., a deficit in effector T cells, or a disequilibrium in the respective numbers of its inflammatory and regulatory macrophages – or, more specifically, of its macrophages distributed along the inflammatory to “alternatively activated” spectrum). Such abnormalities can contribute to explain the triggering of cancer, and they could be targeted by a number of therapies, which precisely aim at correcting these immune defects.
当免疫系统促进癌症进展时,无论是因为消除或遏制癌症的缺陷,还是通过免疫耐受的维护和/或机体修复机制,此时的免疫系统是否存在功能障碍?我建议在这里区分两种情况。第一种情况免疫系统的功能失调。一个生物体,无论是结构上的还是暂时的(例如,在用免疫抑制药物治疗后),都可能有一个有缺陷的免疫系统(例如,效应 T 细胞的缺陷,或其炎症性和调节性巨噬细胞的比例失衡——或者, 更具体地说,它的巨噬细胞处于炎症和“不稳定活跃”的状态)。这种异常有助于解释癌症的触发,并且很多免疫疗法都旨在纠正这些免疫缺陷。
In the second situation, however, the immune system acts normally and immune-mediated decohesion is due to an abnormal context. Pathogens, wounds, mechanical pressure, and local modifications due to carcinogenic environmental factors, among several other causes, can create an abnormal local context (characterized by inflammation, perturbation of the extracellular matrix, and so on). This local context influences the immune system, which in turn responds as it usually does, that is, by maintaining or repairing the tissue – even if the final, pathological, outcome is cancer promotion (Figure 4.4). It has long been known, for instance, that tumors resemble “wounds that do not heal”, which means that, in cancer, normal repair mechanisms are triggered but generally without reaching the “resolution phase” (which, in the physiological context, is indispensable to terminate the reparative process). Furthermore, the tumor itself can be a major source of perturbation of the local context: it can influence the immune system through a variety of cytokines and can also increase inflammation and wounding, modify blood vessels, reshape the extracellular matrix, or exert a mechanical pressure, among many other possibilities. This is often described as the “hijacking” or “co-option” by the tumor of physiological pathways and of the tissue microenvironment. Despite its importance, one must keep in mind that such co-option is only one of the many ways in which the local context can become abnormal and favor the contribution of immune processes to cancer progression.
然而,在第二种情况下,免疫系统正常运作,免疫介导的脱散(decohesion)是由于异常环境。 病原体、伤口、机械压力和致癌环境因素引起的局部变化,以及其他几个原因,可以产生异常的局部环境(以炎症、细胞外基质的扰动等为特征)。这种局部环境会影响免疫系统,而免疫系统又会像往常一样做出反应,即通过维持或修复组织——即使最终的病理结果是促进癌症发生(图 4.4)。例如,人们早就知道肿瘤类似于“无法愈合的伤口”,这意味着在癌症中,正常的修复机制会被触发,但通常不会达到“消退期”(在生理学背景下,对终止修复过程必不可少)。此外,肿瘤本身可能是局部环境扰动的主要来源:它可以通过多种细胞因子影响免疫系统,还可以增加炎症和损伤、改变血管、重塑细胞外基质或施加机械压力 ,以及许多其他可能性。这通常被描述为肿瘤对生理通路和组织微环境的“劫持”(“hijacking” )or或“共同选择”(“co-option”)。尽管它很重要,但必须记住,这种共同选择只是局部环境可能变得异常、免疫促进癌症进展的众多可能机制之一。
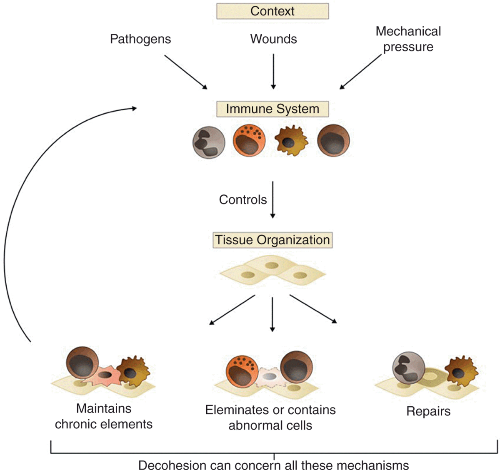
Figure 4.4 Decohesion in cancer induced by an abnormal context. Immune-mediated decohesion may be due to an abnormal context rather than an abnormal immune system. This abnormal context can be due to the presence of pathogens, wounds, mechanical pressures, and carcinogens of environmental origins, among many other resources; it can also be triggered by the tumor itself. In many situations, the decohesion mediated by the immune system results from abnormal realization of normal processes (such as maintenance and repair).
(Figure drawn by Wiebke Bretting).
图 4.4 由异常环境引起的癌症的脱散(decohesion)。免疫介导的脱散可能是由于环境的异常而不是免疫系统的异常。这种异常情况可能是由于存在病原体、伤口、机械压力和环境致癌物以及许多其他资源造成的;它也可以由肿瘤本身触发。在许多情况下,免疫系统介导的脱散是正常生理过程(如维护和修复)的异常实现造成的。
(图由 Wiebke Bretting 绘制)。
In all these contexts, immune-mediated decohesion results from the abnormal realization of normal processes, and this can help explain why tumors largely resemble organs and are the products of classic developmental and reparative pathways realized in an abnormal context. There is no doubt that the outcome is pathological (the promotion of a cancerous tumor, including in some cases metastatic spread), but the immune system, in many of these circumstances, does not strictly speaking “dysfunction”; it just does what it always does (maintaining the local environment, repairing in case of wound, etc.).
在所有这些情况下,免疫介导的脱散都是正常生理过程异常发生的结果,这有助于解释为什么肿瘤在很大程度上类似于器官,并且是经典的发育和修复通路在异常情况下的产物。毫无疑问,结果是病理性的(促进癌性肿瘤,在某些情况下包括转移扩散),但在许多情况下,免疫系统严格来说并没有“功能障碍”;它只是做它一直做的事情(维护局部微环境,在受伤的情况下进行修复等)。
If the view presented here is correct, then a much richer picture emerges about how the immune system influences cancer and, ultimately, of potential therapeutic opportunities as well. Indeed, the immune system influences cancer through different processes (elimination, containment, maintenance, repair, and so on), via many actors (not only lymphocytes, but also macrophages, neutrophils, and various cytokines), at several different levels (within the tumor, but also around the tumor, in the whole tissue, and at a systemic level in the organism), and at all temporal stages of cancer progression (initiation, neoplastic progression, and metastasis). Additionally, the influence of the immune system on the cancerous tumor can be negative (the immune system prevents cancer progression) or positive (the immune system promotes cancer progression).
如果这里提出的观点是正确的,那么关于免疫系统如何影响癌症以及最终潜在的治疗机会的更丰富的画面就会出现。事实上,免疫系统通过不同的过程(消除、遏制、维持、修复等),通过许多参与者(不仅是淋巴细胞,还有巨噬细胞、中性粒细胞和各种细胞因子),在几个不同的水平(肿瘤、肿瘤周围、整个组织和生物体的系统水平),以及癌症进展的所有时间阶段(起始、肿瘤进展和转移)影响癌症。此外,免疫系统对癌性肿瘤的影响可能是抑制的(免疫系统阻止癌症进展)或促成的(免疫系统促进癌症进展)。
All this suggests a whole series of new opportunities for investigating immunotherapies, which could, at least in principle, target these different processes, actors, levels, and temporal stages. Current immunotherapies (particularly immune checkpoint inhibitors and CAR-T cells) focus on lymphocytes in terms of actors and on elimination and maintenance and rupture of chronicity in terms of processes, but many other possibilities exist. Depending on where we are in the cycle of cancer–immune system interactions and on the actors of the tumor microenvironment involved, some therapeutic strategies will aim at normalization while others will aim at denormalization. Examples of normalization include the reduction of the level of inflammation in the tissue, the elimination of pathogens and/or chronic wounds, the restoration of immune accessibility to the tumor, and the facilitation of the resolving phase of tissue repair. In contrast, denormalization would be a major aim when the immune system interacts with tumor components as if they were normal constituents of the body, as, for example, when the immune system is tolerogenic in the context of chronically present tumor antigens or when the immune system continuously triggers repair pathways to respond to a local cancerous context that displays many features usually associated with a wound.
所有这些都表明了研究免疫疗法的一系列新机会,至少在原则上,免疫疗法可以针对这些不同的过程、参与者、水平和时间阶段。当前的免疫疗法(特别是免疫检查点抑制剂和 CAR-T 细胞)在对象上侧重针对淋巴细胞,在过程方面侧重于慢性病的消除、维持和破坏耐受,但是其实免疫疗法但还存在许多其他可能性。根据我们在癌症-免疫系统相互作用的循环中所处的位置以及所涉及的肿瘤微环境的参与者,一些治疗策略将以“正常化”为目标,而另一些则以“去正常化”为目标。“正常化”的例子包括降低组织中的炎症水平、消除病原体和/或慢性伤口、恢复对肿瘤的免疫可及性以及促进组织修复的解决阶段。相反,当免疫系统与肿瘤成分相互作用时,免疫系统视肿瘤为身体的正常成分时,例如,当免疫系统在肿瘤抗原长期存在的情况下具有耐受性时,或者当免疫系统不断触发修复途径以响应呈现伤口相关特征的局部癌变环境。“去正常化”将是肿瘤免疫治疗的一个主要目标。
In summary, this section has shown that focusing on the immune system is essential for anyone studying cancer. Cancer is a disease of multicellularity and, more specifically, of the cohesion of the multicellular organism. Immunological surveillance constitutes one of the main and best described mechanisms by which the multicellular organism exerts control over lower-level entities. A major result of recent research is that the immune system can both restrain and promote cancerous tumors, which may seem, at first sight, paradoxical. Yet the situation becomes less paradoxical when one realizes that immune-mediated decohesion is often due to an abnormal context rather than a dysfunctional immune system. We have suggested here an extended view of cancer–immune interactions, which opens up many opportunities for investigating new mechanisms of tumor control and tumor promotion and, ultimately, for developing novel therapeutic opportunities based on the action of the immune system.
总而言之,本节表明,关注免疫系统对于任何研究癌症的人来说都是必不可少的。癌症是一种多细胞疾病,更具体地说,是多细胞生物体凝聚(cohesion of the multicellular organism)的疾病。免疫监视(immunological surveillance)构成了多细胞生物对低级实体施加控制的主要机制之一,也是描述得最好的机制。最近研究的一个主要结果是免疫系统可以抑制和促进癌性肿瘤,乍一看,这似乎是自相矛盾的。然而,当人们意识到免疫介导的脱散(decohesion)通常是由于异常环境而不是免疫系统的功能失调引起时,情况就变得不那么矛盾了。我们在这里提出了癌症-免疫相互作用的扩展观点,这为研究抑瘤和促瘤的新机制以及开发新的免疫疗法提供了思路。
本文仅作信息分享,不代表礼进生物公司立场和观点,也不作治疗方案推荐和介绍。如有需求,请咨询和联系正规医疗机构。
参考文献1. Pradeu, T., Philosophy of Immunology. 2020, Cambridge University Press: Cambridge.




 沪公网安备 31011502015333号
沪公网安备 31011502015333号